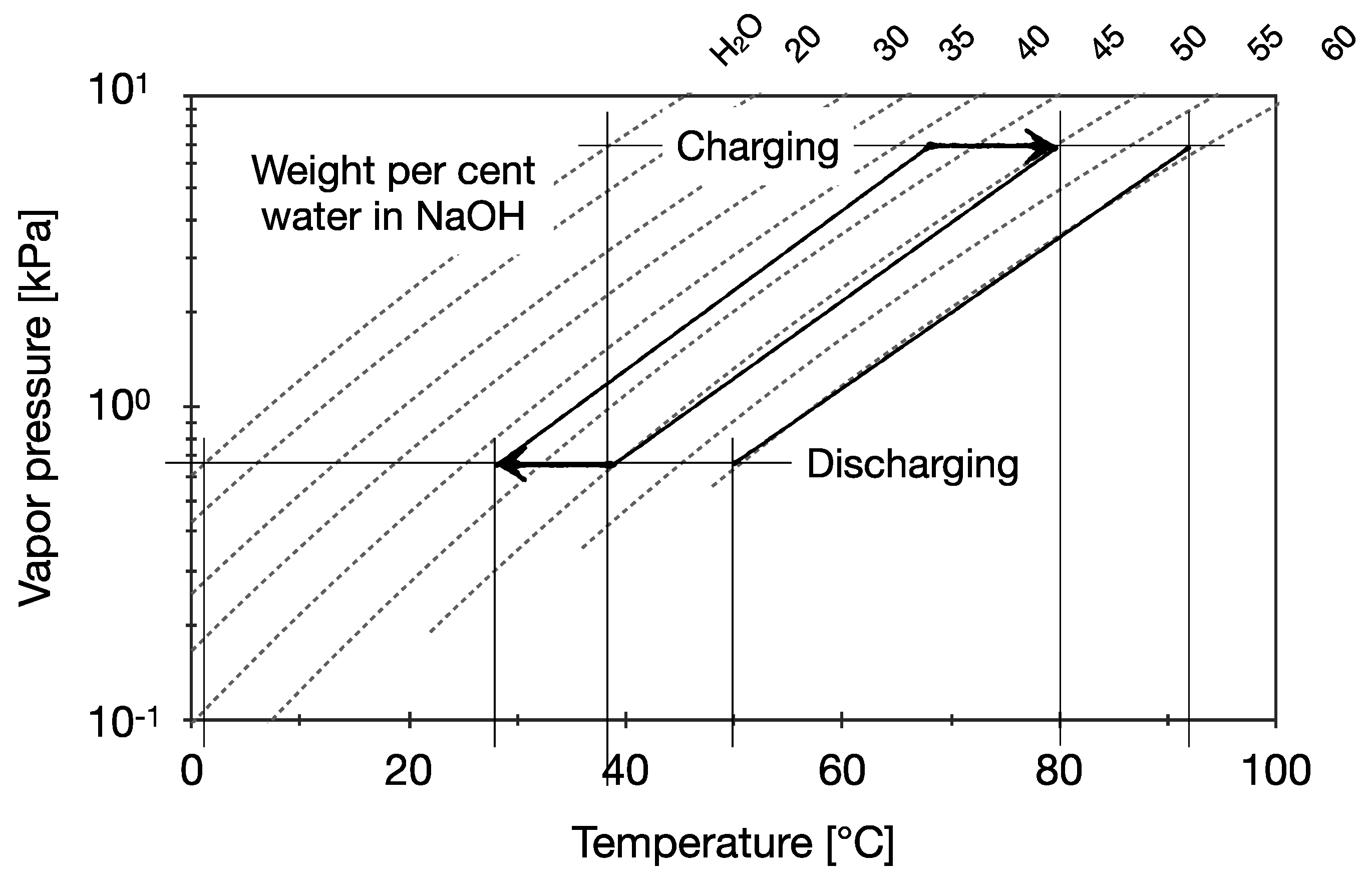
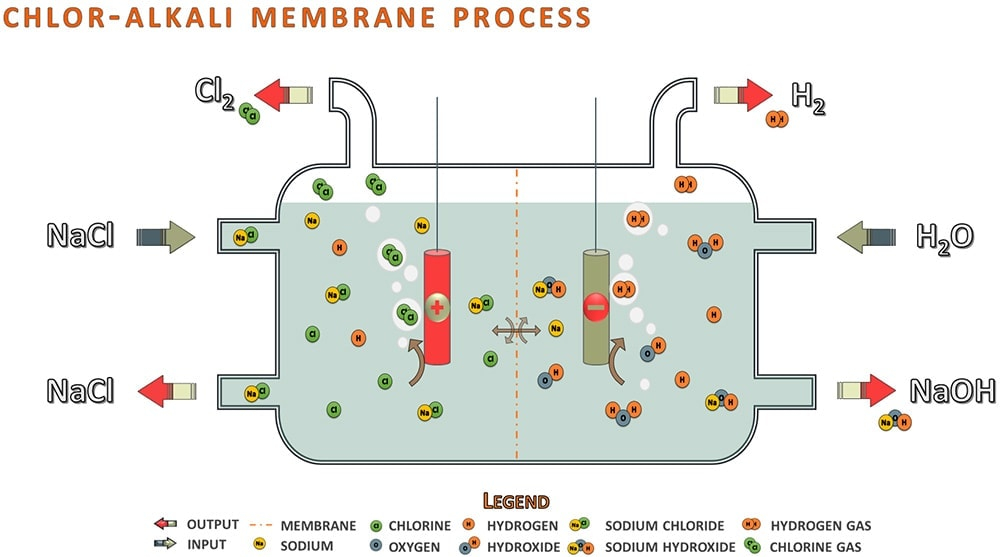
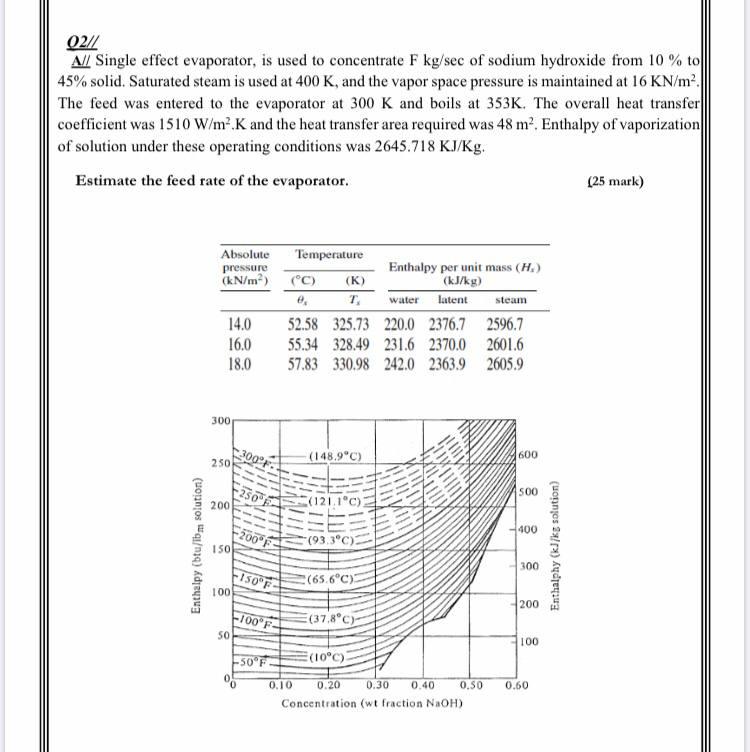
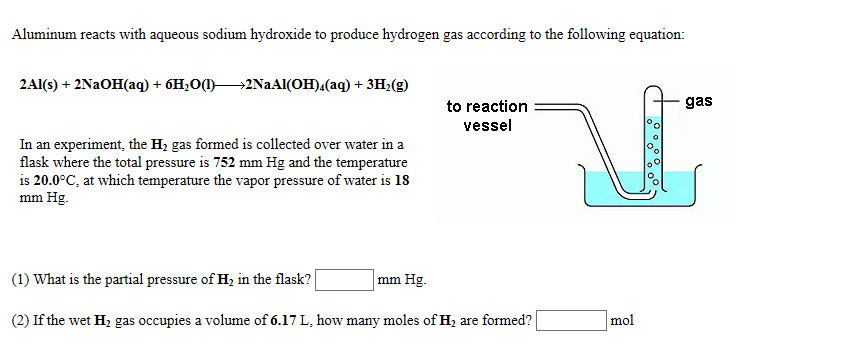
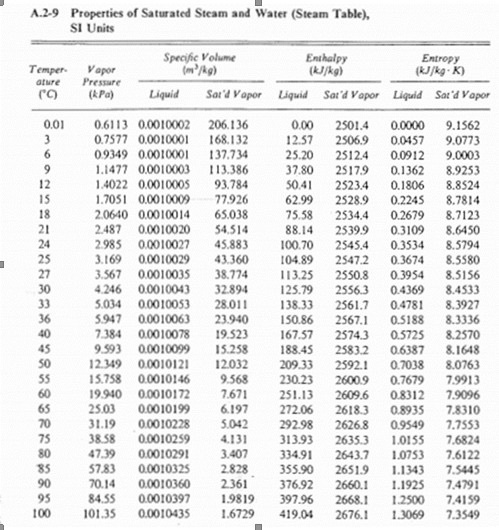
Title P-V-T relations of aqueous sodium hydroxide and its causticity at high temperatures and high pressures Author(s) Kiyama, R
![A): Viscosity of aqueous solutions of NaOH (Data from reference [71]),... | Download Scientific Diagram A): Viscosity of aqueous solutions of NaOH (Data from reference [71]),... | Download Scientific Diagram](https://www.researchgate.net/publication/322865826/figure/fig2/AS:645814003195906@1530985465666/A-Viscosity-of-aqueous-solutions-of-NaOH-Data-from-reference-71-KOH-data-from.png)
A): Viscosity of aqueous solutions of NaOH (Data from reference [71]),... | Download Scientific Diagram

SOLVED: Sodium hydride reacts with excess water to produce aqueous sodium hydroxide and hydrogen gas. How many grams of NaH will produce 0.982 L of gas at 301 K and barometric pressure
Title P-V-T relations of aqueous sodium hydroxide and its causticity at high temperatures and high pressures Author(s) Kiyama, R

Predicting the Autoaccelerating Hydrogen Peroxide Decomposition Rate after Mixing with Sodium Hydroxide | Industrial & Engineering Chemistry Research
I P-V-T RELATIONS OF AQUEOUS SODIUM HYDROXIDE AND ITS CAUSTICITY AT HIGH TEMPERATURES AND HIGH PRESSURES BY Rv0 I\IYA]IA AVD SHI

Calculated vapor pressures of K and Na vs. temperature. For K, both... | Download Scientific Diagram
UC-600 Ammonia in Simulated Hanford Double-Shell Tank Wastes: Solubility and Effects on Surface Tension

1: Saturation temperatures for sodium hydroxide solutions for different... | Download Scientific Diagram